Project Updates
January 2023
This webinar provides a description of microorganisms leading to intramammary infections in first-lactation organic dairy cows, their risk factors and their relationship with udder health on the following lactation. The findings of a NIFA OREI funded research project provide important information that could be used to develop tailored mastitis control plans for first-lactation organic dairy cows. Presented by Felipe Peña Mosca of the University of Minnesota
October 2021
Noelle Noyes gave a presentation: "The Latest Microbiome Research" on the Dairy Signal podcast, produced by the Professional Dairy Producers of Wisconsin (PDPW). View the presentation at https://mediasiteconnect.com/site/pdpw-dairy-signal/watch/d1d312c9-1b95-4ec7-efbf-08d989a8c617
September 2021
Click on the Publications section of this website for links to our recently published articles and for researchers, a link to reading materials from an online course on Introduction to statistical analysis of metagenomic sequencing data!
March 2021
We published a new extension article:
"What is the Microbiome and Why is it Important for Organic Livestock Production?"
This article can be accessed here
March 2020
Noelle Noyes gave a webinar about udder microbiome and how it might impact organic dairy production
January 2020
We are almost done with sample collection. We have reached a number of 450 heifers enrolled from 5 organic dairy farms.



April 2019

Summary
April has been a very busy month for all aspects of the project, including sample collection/preparation, ordering supplies, finalizing protocols, and presentations.
Sample Collection Updates
Over the past several months, we have been collecting udder skin samples from heifers in Minnesota and Texas, having enrolled nearly 200 animals in total. Some of these animals have already begun to calve, which means that we are moving into the next stage of sample collection, i.e., the collection of both teat canal and stripped milk samples.
This is exciting for a number of reasons: first, cows are most susceptible to mastitis at freshening, which means that this is the stage where we expect to see the most dramatic changes to the bovine teat microbiome; and second, we will soon be able to perform bacterial culture for mastitis pathogens on the collected milk samples, which means that we will soon begin to see some results!
Sample Processing
Earlier this month we met with program leaders at the University of Minnesota Genomics Core (UMGC) and the Udder Health Lab (UHL) to discuss our microbiome sequencing and culture needs, and decided on the following data processing protocols moving forward:
- DNA extraction will be performed using the MagAttract PowerMicrobiome DNA Kit
- Amplification and sequencing will be performed by the UMGC on the V4 region of the 16S rRNA gene
- Bacterial culture for mastitis pathogens (Staph spp., Strep spp., Prototheca spp.) will be conducted by the Udder Health Lab at the University of Minnesota
Presentations
Chris Dean -- leading Ph.D. student on the project -- recently gave two presentations, introducing the Open-ROAMER project. The first was to a room of excited graduate students and faculty during the bi-weekly Quantitative Epidemiology Seminar at the University of Minnesota, and the second was at the 2019 Minnesota Dairy Health Conference at the University of Minnesota. Way to go Chris!
January 2019


Summary
One of the great things about interdisciplinary projects like this one is that it affords the opportunity to work with a "real team", i.e., a collection of individuals with a diverse set of skills that come together for the purpose of addressing a common goal.
Naturally, this means that people will be doing some things that they have not done before, e.g., visiting an organic dairy, collecting samples, or interacting with key stakeholders.
Therefore, the main purpose for the month of January was to give members of our team -- those without a formal background in dairy production -- a chance to visit an organic dairy, design and execute their own sample collection experiment, and interact with local Minnesotoan farm producers.
December 2018
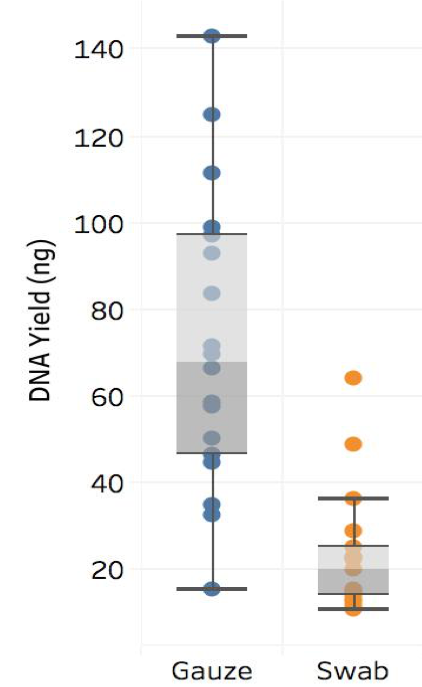
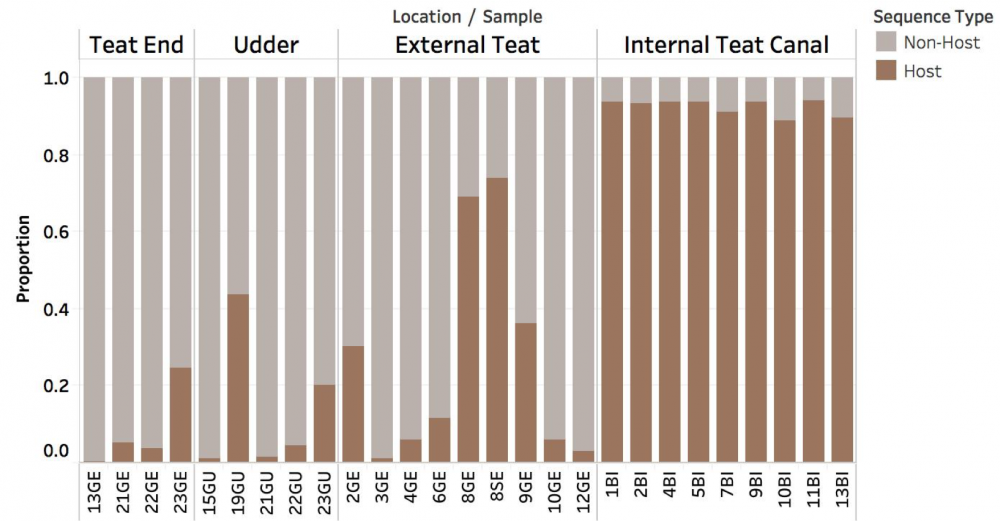
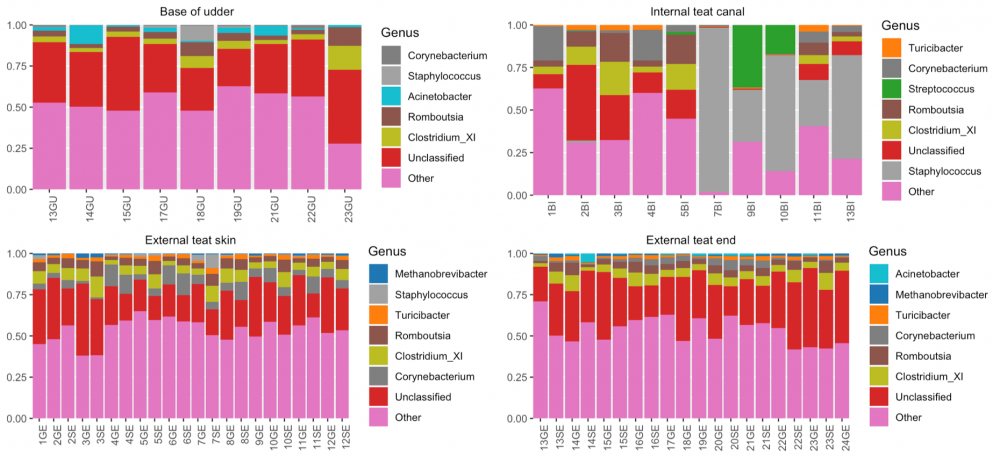
Summary
In July, we conducted a pilot study with three goals in mind: the first, to optimize a sampling protocol for the isolation and recovery of DNA from various regions of the cow udder epithelium; the second, describe the microbial diversity of the cow udder epithelium; and third, to quantify the proportion of host versus microbial DNA that can be expected from these samples.
Results
Overall, we found that gauze squares yielded significantly more DNA than cotton tip swabs (left figure). We also found that the proportion of host versus non-host DNA was highly variable between the different sampling locations (middle figure). Notably, we saw that the majority of host-derived sequences could be isolated from the internal teat canal, whereas the teat end showed the least amount of host. In terms of relative abundance, we found that the cow udder microbiome is composed of a variety of microorganisms -- some of which are more abundant in some regions of the cow udder than other -- including Staphylococcus spp., Streptococcus spp., Corynebacterium spp., and many others (right figure).
Dissemination of Results
These results -- and others -- are currently being prepared for submission to a peer-reviewed journal.
July 2018

Summary
We recently received funding from the USDA -- through their Organic Research and Extension Initiative -- to conduct a very large-scale, prospective, epidemiological investigation into the cow udder microbiome and potential associations with mastitis.
To measure the exposure (the microbiome), cow udder epithelium samples will be collected, weekly from 600 heifers, beginning 8 weeks pre-partum, and ending 4 weeks post-partum. To measure the outcome (mastitis), stripped milk samples will be collected weekly at beginning at freshening and ending 4 weeks into early lactation.
Before undertaking such a massive sampling campaign, it is important that we optimize a sampling protocol for the isolation and recovery of bacterial DNA. This is important because we will be collecting skin samples, i.e., samples with a small amount of microbial biomass, so it is critical that we identify a sample collection instrument that will yield the most amount of genomic material that we can work with moving forward.
To do this, project PIs (Drs. Noelle Noyes and Luciano Caixeta), along with two graduate students (Kathryn Crone and Ilya Slizovskiy) conducted a pilot study at a local organic dairy in Minnesota.
Objectives
The main objectives of this pilot study were three-fold:
- To optimize a sampling protocol for the isolation and recovery of bacterial DNA from the cow udder epithelium.
- Determine the composition of the cow udder microbiome from distinct locations of the cow udder (base of the udder, external teat skin, teat end and teat canal).
- Quantify the proportion of bovine versus microbial DNA originating from these locations.
Study Design
To address the objectives of this study, 24 organic dairy cattle were enrolled. In total, 72 samples were collected (three from each cow) using a combination of sample collection devices (gauze square, cotton-tip swab, cytobrush swab). These were stored on ice and brought back to the University of Minnesota, where they will await DNA extraction, 16S rRNA amplification, and next-generation sequencing.
Results
Stay tuned!
Funding

This project is funded by the Organic Research and Extension Initiative grant, part of the USDA National Institute of Food and Agriculture. Grant number 2018-51300-28563

